Researchers at the MRC Protein Phosphorylation Unit and the Centre for Virus Research at the University of Glasgow have uncovered a previously unknown antiviral mechanism that functions like a molecular "quarantine" for viruses. The study, published in Molecular Cell, reveals how a protein called ZNFX1 senses viral RNA and uses a unique form of ubiquitin circuitry to entrap and neutralize the threat, at least transiently.
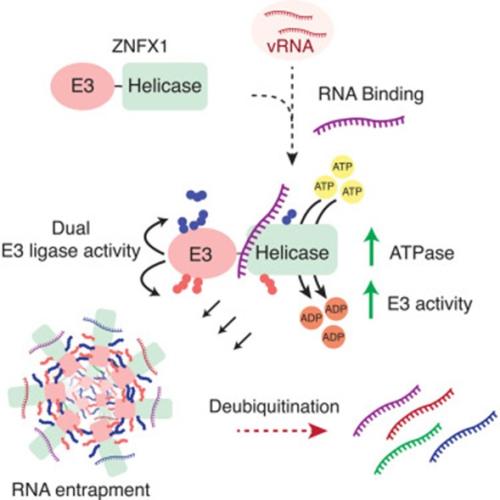
While most proteins have one specific job, the team discovered that ZNFX1 is a rare "mechanistic hybrid." It functions both as an RNA helicase and an unconventional E3 ubiquitin ligase with bimodal activity.
This is the first time an immune factor has been found to combine these two specific activities in a single polypeptide. By using ATP as fuel to bind to viral RNA, ZNFX1 triggers a "two-component ubiquitin circuit." This circuit works in parallel, triggering two different ubiquitin molecular signals: one that destroys the protein, and another that acts as "glue."
The research team found that when ZNFX1 detects a virus, it begins to form self-propagating aggregates – large, stable clusters that effectively "web" the viral RNA in place. This entrapment puts the brakes on virus replication, by putting its genetic material into a molecular quarantine.
"ZNFX1 coordinates a direct antiviral response by creating a 'web of ubiquitin' that drives the assembly of these traps," explains Professor Satpal Virdee, one of the lead authors and founder of Outrun Therapeutics. "Remarkably, we found that this process is reversible. The traps can be dissolved by deubiquitinating enzymes, showing that this is a highly controlled and sophisticated defence system. These findings pave the way to new antivirals and autoinflammatory drugs that work by altering the duration of RNA lock up – sometimes we might want to throw away the key, others a reduced sentence may be preferable."
The study also provides an explanation for why mutations in the ZNFX1 gene lead to severe paediatric immunodeficiencies. The researchers showed that these mutations impair viral restriction in children's immune systems, presumably due to an inability to effectively trap and restrict viruses like Sars.
Dr Adam Fletcher, UKRI Future Leadership Senior Fellow at the CVR, who co-led the study, said: “This is yet another elaborate way that the cell defends itself from viruses. By temporarily entrapping viral genomes, ZNFX1 seems to act like a ‘speed bump’ for the virus. Understanding how these antiviral webs form and why they dissolve would help us figure out whether they can be harnessed as antiviral medicines in the future”.
Understanding this "molecular trap" opens new doors for therapeutic strategies. By learning how to tune this ubiquitin-mediated RNA silencing, scientists may be able to develop new host-directed treatments for chronic viral infections and autoinflammatory diseases.
"This discovery shifts our understanding of how the body responds to viral infection. By acting as both a sensor of viral nucleic acids and an effector of antiviral signalling, ZNFX1 bridges the gap between metabolism, motor activity and the ubiquitin system to stifle viral replication before traditional immune pathways have even kicked in." - Dan Squair, co-first author

